The characterization of materials has a fundamental role to play in the advancement of science and technology. Among the various techniques used to analyze material properties, Photoluminescence Spectroscopy stands out for its ability to study the photophysical and luminescent properties of different materials. This technique is widely used in fields such as semiconductor research, the study of nanomaterials, and the exploration of organic compounds. It provides valuable information on the structure and behaviour of materials at the atomic and molecular levels.
In the field of semiconductors, photoluminescence spectroscopy has been a key method for investigating crystal quality and defect properties in semiconductor structures. It has also been widely used to characterize nanomaterials, to understand their unique luminescence properties and to explore their potential applications in electronics and optoelectronics. In the field of organic materials science, this technique has been essential for investigating the optical properties of polymers and small molecules, and for optimizing photovoltaic and OLED devices.
Exploring Photoluminescence Spectroscopy
Photoluminescence spectroscopy is a non-destructive technique that studies the emission of photons from a material after it has been excited by light of a specific wavelength. As shown in Figure 1, the process begins with the absorption of incident light by electrons in the material. This causes them to transition to higher energy states. These electrons then return to their original energy state, releasing photons whose energy is related to the energy bands and electronic structure of the material.
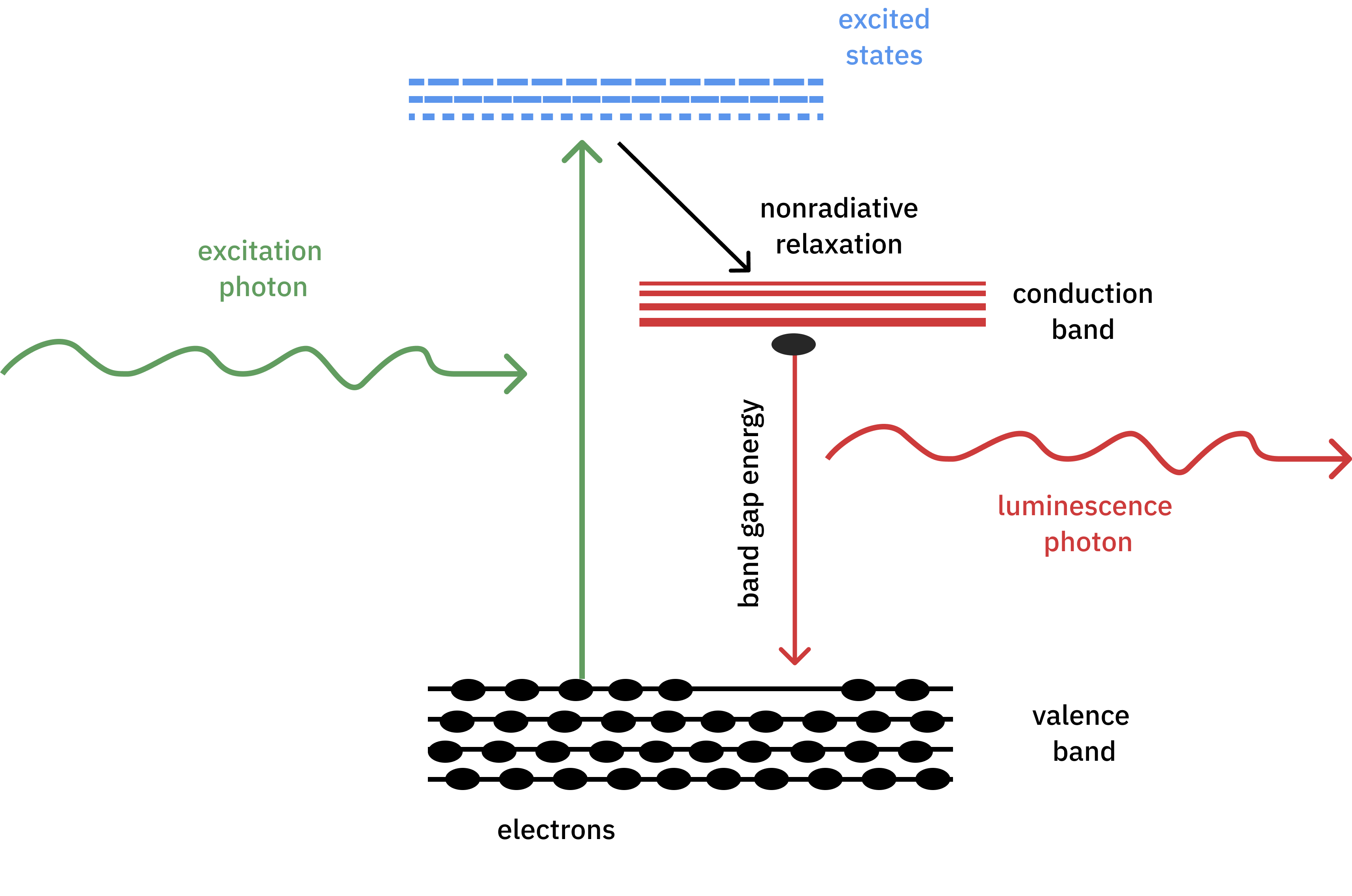
Photoluminescence spectroscopy provides valuable information about the band structure, the fluorescence half-life, and the electronic transitions of a material. This information is crucial to understanding the optical and electronic properties of materials, which in turn opens opportunities to improve electronic devices and develop new materials for various applications.
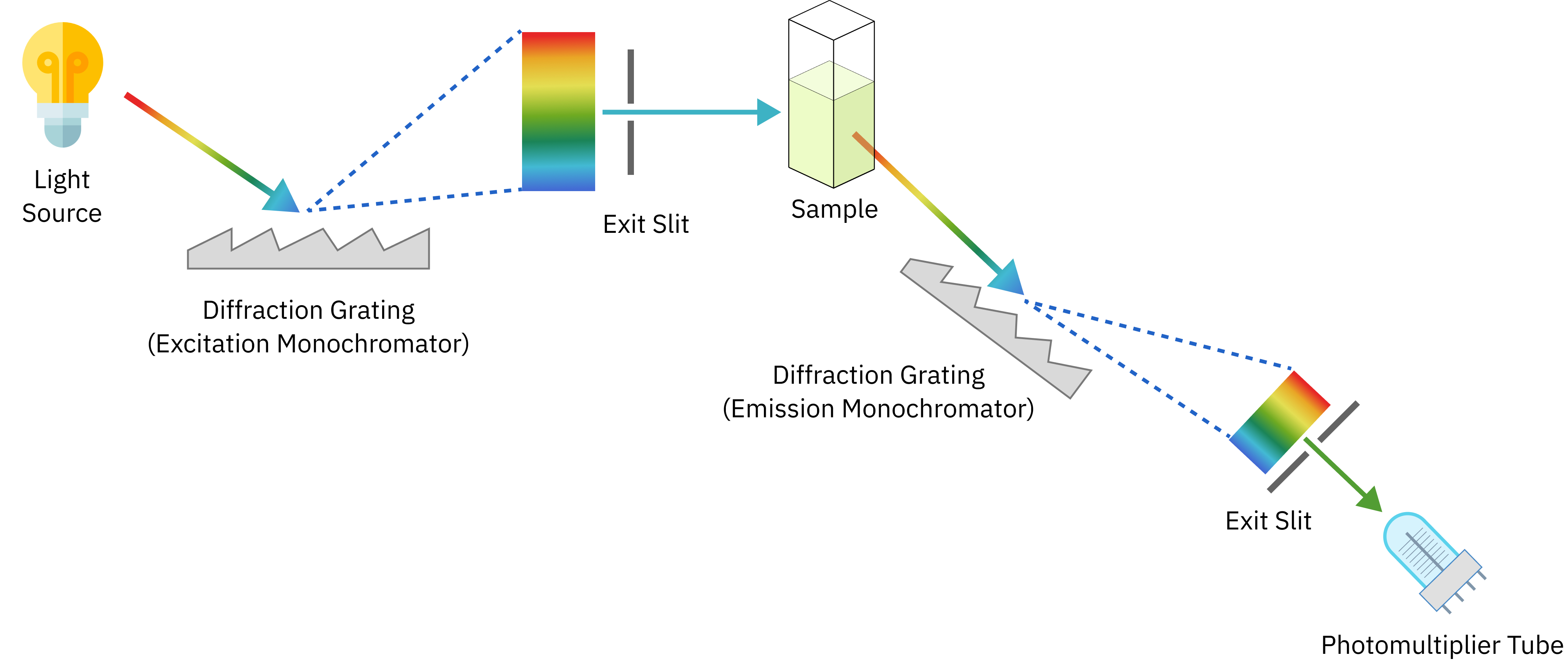
Typically, photoluminescence spectroscopy is performed in the laboratory using a homemade spectrofluorometer (or Photoluminescence Spectrometer) equipped with an excitation section (light source + excitation monochromator), the sample, and an emission section (emission monochromator + detector).
The sample is exposed to the excitation light and the photon emission produced by the sample is measured. The data obtained from the resulting spectrum is used to identify the photophysical and luminescent properties of the material under investigation.
Limitations of Current Light Sources in Photoluminescence Spectroscopy
As mentioned above, this technique is commonly employed for characterizing materials, but the conventional light sources used in this technique have certain limitations.
Xenon arc lamps, commonly used in spectrometers, provide a wide range of excitation wavelengths, but their emission consists of discrete lines, limiting the precise selection of wavelengths for certain materials with specific optical transitions.
Single-wavelength lasers, on the other hand, provide highly coherent light at a specific wavelength. While they are ideal for applications requiring precise excitation, their use may be limited to materials with absorption bands that match the wavelength of the laser.
In addition, the response speed of the xenon arc lamp may not be sufficient to capture ultrafast fluorescence processes, while some single-wavelength lasers can offer similar speeds but are limited by their fixed wavelength.
These limitations can affect the ability of researchers to fully characterize the luminescence of certain materials and understand their photophysical properties in detail. Advances in technology have therefore led to the development of the supercontinuum as a promising solution to improve the characterization of materials in photoluminescence spectroscopy and open new opportunities in scientific and technological research.
Our Supercontinuum Laser Solution: FYLA Iceblink
The Iceblink’s specifications make it one of the best solutions for solving the limitations that common light sources have in photoluminescence spectroscopy.
- Broad spectral range: While conventional light sources have a limited wavelength range, Iceblink offers a spectrum from 450 to 2300 nm. This broad coverage allows precise selection of the right wavelength to excite different materials, overcoming the limitation of selecting specific wavelengths. It also permits the study of a single unique material over a wide range of spectral ranges, resulting in a greater understanding of that material.
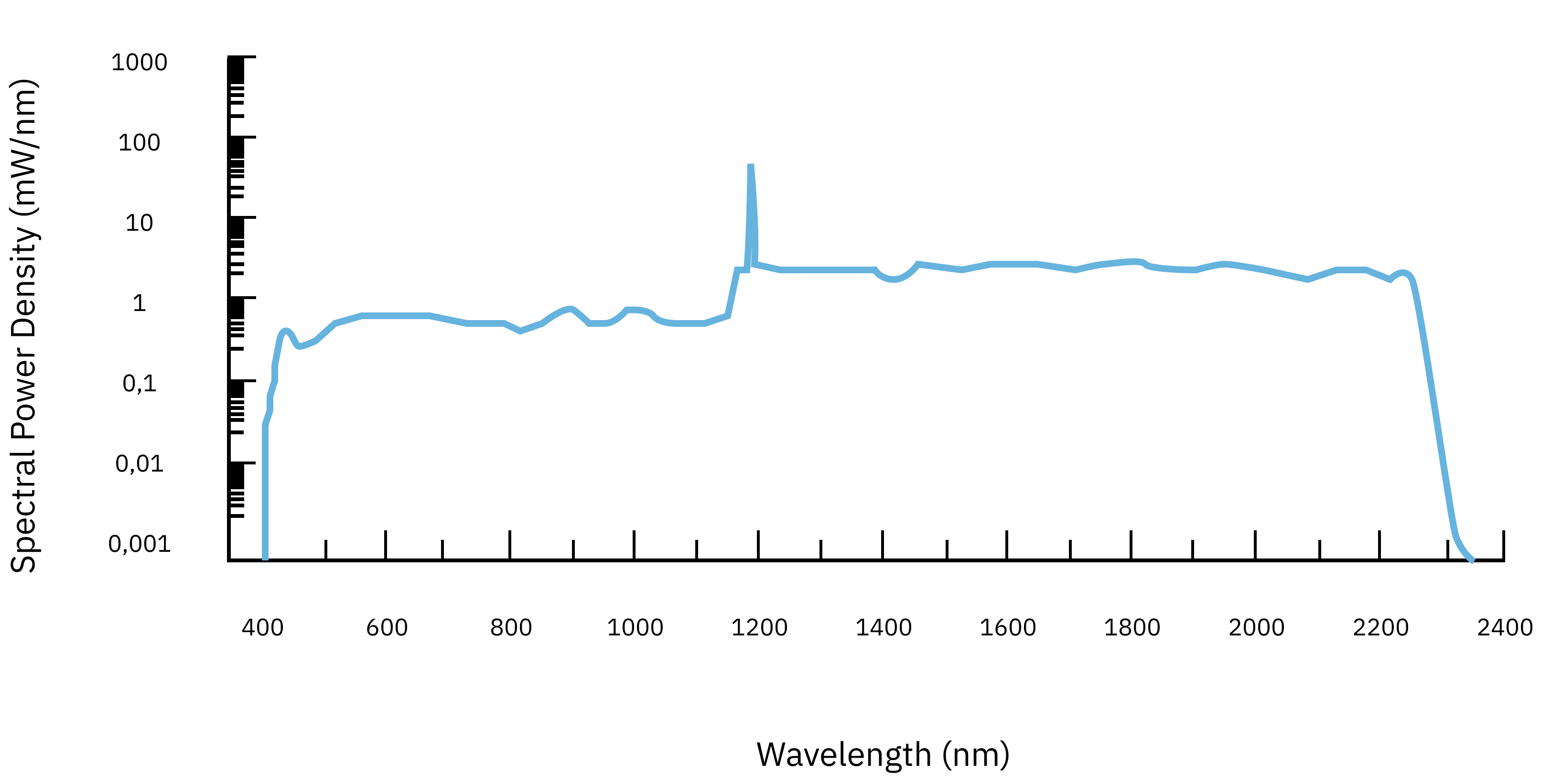
- High Power and Excellent Stability: Iceblink offers a total power of > 3 W. This high power ensures efficient excitation, improving the detection of weak signals and enabling accurate characterization of materials with low luminescence efficiency. In addition, the high-power stability, with a standard deviation of < 0.5% (std. dev.), ensures reliable and consistent measurements over time, avoiding the fluctuations observed with conventional light sources.
- Ultrashort Pulses and High Repetition Rate: With pulses of < 10 ps at the fundamental wavelength and a repetition rate of 80 MHz, Iceblink is suited to capturing ultra-fast luminescence events. This feature overcomes the response speed limitation of conventional light sources, allowing photophysical processes to be studied on the picosecond timescales with outstanding temporal resolution.
In short, Iceblink solves the limitations of conventional light sources by providing a wide spectral range for accurate excitation, intense and stable power for reliable characterization, and ultra-short pulses with high repetition rates to capture ultra-fast luminescence processes. These features make Iceblink an exceptional tool for the application of photoluminescence spectroscopy, enabling researchers to perform detailed studies of luminescent materials.