Diamonds are one of the most interesting materials that are used nowadays for technological applications. The rising interest and development of new techniques to produce diamonds, and their attractive characteristics such as wide band gap, transparency in the whole VIS to NIR spectrum and their high thermal conductivity, contribute to their practical usage. [1]
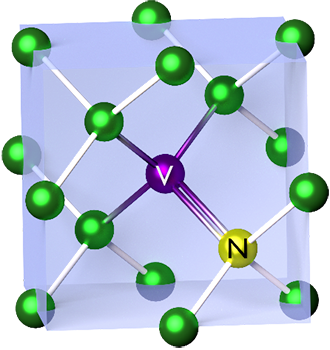
In this scope, the manipulation of crystal diamonds has led to the discovery of color centers. Color centers in Single-crystal diamonds are induced point defects that can absorb and emit light, leading to the appearance of color in the material. One of these defects consists of the substitution of a pair of carbon atoms for a nitrogen atom and an empty space in the diamond lattice, also referred to as lattice vacancy (Figure 1). This type of defect is called Nitrogen – Vacancy center (NV center).
NV centers have a quantum nature that provides them with high sensitivity for external variations of different factors: electric or magnetic field, temperature or strain. They are extremely small, making them suitable for their usage as probes in biological applications and because they are embedded within the structure of the diamond, they are highly stable sensors. [2]
Optical and spin properties of NV Centers and their use for Quantum Information Applications
Nowadays, Nitrogen-Vacancy centers find great use in quantum sensing and quantum computing applications. [3].
In particular, their metrology capacity is related to the quantum mechanical characteristics of the spin state of the impurity.
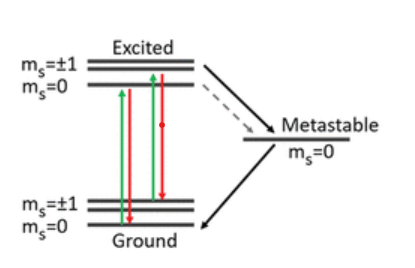
NV centers can be found in two states: neutral state (NV0) or negative state (NV-), nevertheless, only the negative state has technical importance in the field of quantum optics and sensing due to its specific optical and spin characteristics. A typical NV structure contains three electronic energy levels: ground state, excited state and metastable singlet state (Figure 2). The ground and excited states consist of spin triplets levels, that can be polarized by an incident light beam, and can produce broadband photoluminescence emission (Figure 2) [2].
The luminescence emitted from the spin-triplet ground level is specifically interesting due to the great temporal stability of the relaxation process, compared to the traditional single-emitters which tend to be photobleached at room temperatures. Room-temperature quantum entanglement between two defect spins in the NV lattice energy structure with long relaxation lifetimes could be a major contribution to quantum computing [5]. Furthermore, the weak interactions between NV centers and the rest of the atoms in the lattice ensure highly stable emission, which is also a very desirable characteristic for applications related to labelling biological tissues or surface characterizations such as fluorescence. [4]
Iceblink for the excitation of NV centers
Laser light sources with emission in the VIS range and confocal microscopes can be used to excite the NV Centers and study their photoluminescence and fluorescence emission. The most common light sources used for NV excitation include lasers emitting mainly in the green part of the VIS spectrum, such that the emission at red-shifted wavelengths can be studied.
In this scope, we propose the use of the Iceblink supercontinuum laser for the excitation of the NV centers:
- Simultaneous excitation of multiple NV centers with the broadband emission of the Iceblink.
- Combine our Iceblink supercontinuum laser with its Boreal tunable filter to choose excitation wavelengths in the VIS range with a minimum resolution of 10 nm.
- Get the most stable fluorescence intensity measurements with the outstanding power stability of the Iceblink.
- Use the short pulses of <10 picoseconds and the high repetition rate of 80 MHz for a higher sensitivity.
References
[1] Ismagilov R, Malykhin S, Puzyr A, Loginov A, Kleshch V, Obraztsov A. Single-Crystal Diamond Needle Fabrication Using Hot-Filament Chemical Vapor Deposition. Materials (Basel). 2021 Apr 29;14(9):2320. doi: 10.3390/ma14092320. PMID: 33947061; PMCID: PMC8124821.
[2] Schirhagl, Romana and Chang, Kevin and Loretz, Michael and Degen, Christian L , Nitrogen-Vacancy Centers in Diamond: Nanoscale Sensors for Physics and Biology, Annual Review of Physical Chemistry , vol 65, 1, p. 83-105, 2014. DOI: 10.1146/annurev-physchem-040513-103659, PMID: 24274702.
[3] Park, H., Lee, J., Han, S. et al. Decoherence of nitrogen-vacancy spin ensembles in a nitrogen electron-nuclear spin bath in diamond. npj Quantum Inf 8, 95 (2022). https://doi.org/10.1038/s41534-022-00605-4.
[4] A Mzyk, A Sigaeva, and R Schirhagl. Relaxometry with Nitrogen Vacancy (NV) Centers in Diamond. Accounts of Chemical Research 2022 55 (24), 3572-3580. DOI: 10.1021/acs.accounts.2c00520
[5]- Ma Y, Chen J, Wang C. Growth of Diamond Thin Film and Creation of NV Centers [Internet]. Applications and Use of Diamond. IntechOpen; 2023. Available from: http://dx.doi.org/10.5772/intechopen.108159